Elmiron is a prescription drug for treating interstitial cystitis (IC), also known as bladder pain syndrome. The drug, which Janssen Pharmaceuticals manufactures, was first approved by the Food and Drug Administration for use in the US in 1996. It was a relief for patients and clinicians treating interstitial cystitis- a chronic condition affecting the bladder. In 2018, studies from eye patients linked the prolonged use of Elmiron with unique damage to the retina that can result in permanent eye damage, loss of vision, and blindness.
In January 2020, affected persons filed the first Elmiron lawsuit against Janssen Pharmaceuticals. In this case, the plaintiffs alleged that the manufacturer failed to disclose the severe side effects for prolonged Elmiron users. Claiming that using Elmiron caused severe unexpected eye damage, which can lead to impaired vision and blindness. Also, it is alleged that the company may have been aware of the drug side effects and withheld the information from the public and the FDA.
Over one thousand drug users have filed lawsuits against the manufacturer, claiming that pentosan polysulfate sodium contained in Elmiron led to their eyesight problems.
May 2023 confidential settlements
Janssen Pharmaceuticals has embarked on confidential settlements on most of the Elmiron lawsuits. These settlements have reduced the number of new cases filed into the MDL. There is, however, an ongoing battle over payment for the plaintiff leadership committed. The PLC attorneys demand a certain percentage hold back from the paid lawyers’ fees. The amount proposed appears to be excessive to some attorneys.
Elmiron lawsuits progress
As of March 2023, Janssen Pharmaceuticals faced 1,879 Elmiron product liability cases. However, there has been no trial yet. The first lawsuit against Elmiron manufacturer, the company, was filed in 2020.
The plaintiffs in the three preliminary cases were Jeanette Milburn, Valarie Hull, and Kimberly Pelczar. These three alleged that their long-term use of Elmiron contributed to severe sight problems, including pigmentary maculopathy, retinal injury, retinal haemorrhage, and vision degradation. They claimed that Elmiron was defective and blamed the company for not disclosing the drug’s side effects to doctors and the public.
Bellwether cases in Elmiron lawsuit based on CMO No. 17
The Elmiron lawsuits have been consolidated into a class action MDL in New Jersey. Bellwether trials for these cases are likely to start in April 2023. The protocol for selecting the trial cases, major discoveries, and deadlines follows the Case Management Order in the Elmiron MDL (CMO No. 17).
According to the CMO section III, Elmiron bellwether cases were to be selected based on their eligibility and standard requirements, such as documented proof and a complete plaintiff factsheet.
Also, the pool “Bellwether discover cases” would include 20 cases. Half of these cases were selected by the plaintiff’s executive committee (PEC) and the other half by the Lead Counsel for the Defendants. The CMO also instructed these parties to notify the judge of their case selection by 11/22/21.
After selecting the 20 bellwether cases, the plaintiff’s executive committee PEC and the Lead Counsel for Defendants would begin conducting fact discovery. The process entailed standard civic discovery under federal rules, such as requests for documents and depositions.
According to the discovery process guidelines in CMO 17, each plaintiff was to provide two deposition dates in January 2021 by 12/8/21. Attorneys for both sides were to devise a plan for deposing all treating and prescribing doctors between February and March 2022.
The fact discovery for the bellwether cases was to be completed by March 30, 2022, and three cases would be selected as the Bellwether Trial Cases. The court, PEC, and Defendants’ Lead Counsel would select the three. The selection would be followed by expert witness discovery, dispositive motions, and Daubert motions which would take place until the end of September 2022.
The first Bellwether trial was scheduled for 3/27/2023, New Jersey, with the 2nd and 3rd trial dates to be determined.
The presiding Judge, Martinotti, ruled that Maria Windham’s case would remain the first bellwether case. The determination of the Elmiron Bellwether trials is critical as they set the stage for the other cases.
Who qualifies for the Elmiron lawsuit?
If you suspect the long-term use of Elmiron may have affected your eyesight, you might be eligible for compensation. Requirements for pursuing a lawsuit may include the following:
- Used the Elmiron for at least two years
- Experienced Elmiron vision-related symptoms that occurred after January 1, 2010.
- Having been diagnosed with retinal maculopathy, macular degeneration, or pigmentary maculopathy while taking Elmiron or 12 months after discontinuing the medication.
What is Elmiron?
The Elmiron oral drug contains pentosan polysulfate sodium as the active ingredient and is available in 100mg capsules. Pentosan polysulfate is an anticoagulant. Drugs in this category are often used as blood thinners or anticoagulants.
Other ingredients in the drug include:
- Microcrystalline cellulose
- Magnesium stearate
- Synthetic black iron oxide
- Pharmaceutical glaze (modified) in SD-45
- D&C Yellow NO.10 aluminum lake
- FD&C Blue NO. 1 aluminum lake
- FD&C Red NO. 40 aluminum lake
- FD&C Blue No. 2 aluminum lake
- SDA-3A alcohol
- N-butyl alcohol
- Propylene glycol
- Titanium dioxide
What is interstitial cystitis (IC)?
IC is a chronic condition that increases the pressure in the bladder, causing pelvic pain and frequent urination. The disease, which is more common in women, affects about 12% (1.2M) women and 83000 men or in the united states.
In a healthy person, the bladder stretches to accommodate a certain amount of urine. Once it is filled up, a reflex mechanism (through stretch receptors) sends a signal to the brain’s cerebral cortex and thalamus to initiate voluntary control of the urethra sphincter muscles. The individual is thus able to pass standard amounts of urine at the expected frequency.
Those suffering from the disease have problems with signal delivery and thus feel the urge to urinate more frequently, passing smaller amounts of urine. Some of the known causes of the condition include:
- Presence of certain substances in the urine
- Vascular disease
- Problems in the lining of the urinary bladder
- Overstretching of the bladder or trauma
- Genetic factors
- Certain allergies
- Mast cellular abnormalities
- Pelvic floor muscle dysfunction
- Autoimmune disorders
Also, those suffering from interstitial cystitis may show the following symptoms:
- Urgent frequent urination
- Chronic pelvic pain
- Pain in the lower abdomen and lower back
- Painful urination
- Pain during sexual intercourse
- Pain in the testicles and penis in men
- Pain in the perineum and urethra for women
How does Elmiron work in the treatment of IC?
In most patients with IC, the condition occurs from the presence of abnormal elements in the urine that damage the mucus lining of the bladder. The drug adheres to the bladder wall, protecting it against irritation by these compounds in the urine. Although the medication doesn’t cure the condition, it reduces the symptoms, such as inflammation and pelvic pain.
There currently are no generic drug alternatives for Elmiron to treat interstitial cystitis. However, doctors may prescribe NSAIDS and tricyclic antidepressants for pain relief or antihistamines.
Studies related to Elmiron eye damage
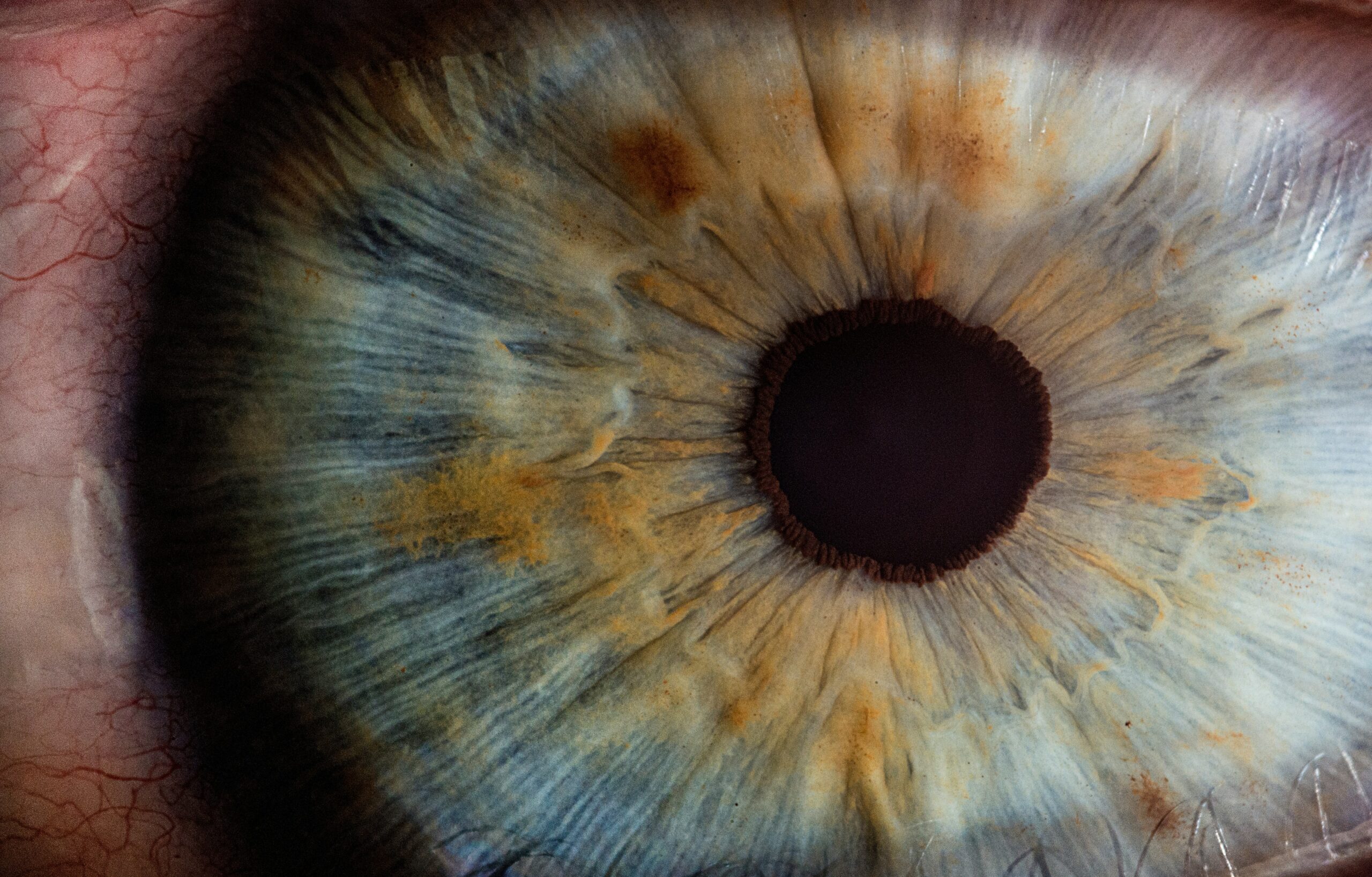
It was only in 2018 that researchers discovered the connection between eye damage and the use of Elmiron. Dr. Joseph T Nezgoda and Dr. Nieraj Jain from Emory eye center, Atlanta, noted that six patients with dystrophy had been using Elmiron for around 16 years. Studies carried out on these patients were recorded in the November 2018 issue of Ophthalmology, pointing to the link between the drug and pigmentary maculopathy.
Emory eye center then wrote to the Journal of Urology, reporting the findings of this unusual retinal pigmentary changes or maculopathy following the prolonged Elmiron use. The patients involved in the study did not have a family history of such a condition and had been using the drug for an average of 15.5 years. The youngest patient in the study was the most severely affected. He had started using Elmiron at 23 and began showing symptoms at 30 years; by 37 years, his condition had worsened.
Another study by Kaiser Permanente ophthalmologists (Drs. Robin A. Vora, Amar P. Patel, and Ronald Melles) on Elmiron use and eye damage indicated that 25% of the drug users suffered severe eye damage. The study involved an analysis of 4.3 million patients, and out of this number, they found 140 patients who took 5,000 Elmiron capsules for 15 years. They invited 91 patients for examination and discovered that 22 showed signs of drug toxicity and eye damage.
These studies were backed up by others conducted by the American Academy of Ophthalmology in October 2019, indicating that the drug had clear signs of toxicity. A 2022 review of the literature surrounding the use of Elmiron concluded that high doses of pentosan cause maculopathy. The symptoms of the resulting condition are consistent with age-related macular degeneration or rod dysfunction.
Following these discoveries in January 2020, Janssen Pharmaceuticals changed its drug label to include a warning that it can lead to changes in the retina of the eye (pigmentary maculopathy). They also changed the drug packaging to a black box, the most severe pharmaceutical warning label. However, the drug is still on sale, and no recalls have been made.
A publication of an Elmiron use case study by a Harvard researcher in November 2019 reported that the effects of Elmiron continued affecting patients even six years after discontinuing use. In July 2020, PPS-associated maculopathy, a publication by several institutions, confirmed the same drug toxicity. Nine of the 11 patients involved had worsening symptoms, with the affected areas on the retina expanding and atrophy encroaching on the foveal center.
Also, after these discoveries, Elmiron users filed lawsuits against the manufacturing company.
What is Pigmentary maculopathy?
Pigmentary maculopathy is a progressive degeneration of the eye macula region. Macula is the central part of the retina, about 5mm. It is central to the vision and responsible for defining the fine details of an image and color vision. Pigmentary maculopathy symptoms include:
- Trouble seeing things up close.
- Difficulty adapting to dim lighting.
- In severe cases, it can lead to permanent loss of sight or b
- Eye pain.
- Difficulty reading.
- Vision disturbances or distorted vision.
The leading cause of this condition is age, also known as age-related macular degeneration, underlying health conditions such as myopia, secondary macular pucker, diabetes, drug intake, and genetic factors.
There is no cure for Pigmentary maculopathy. However, in some cases, medication such as ranibizumab (Lucentis) and aflibercept (Eylea) can be given to slow the disease’s progression and improve vision. Diabetic maculopathy is managed through the injection of VEGF.
How long does Elmiron take to result in vision problems?
According to Emory Eye Centre studies, users start experiencing sight problems three years after using the drug. However, the average period is fourteen and a half years. Patients in the study showed eye problem symptoms that had lasted for up to nine years.
Symptoms of those affected by Elmiron
The use of Elmiron is alleged to result in three conditions: pigmentary maculopathy, macular degeneration, and retinal maculopathy. Pigmentary maculopathy, in particular, is alleged to occur from using pentosan polysulfate sodium, a component in Elmiron.
Reports from the Emory Eye Centre study also indicated that some of the patients affected by Elmiron had shared symptoms, which vary from mild to severe:
- Taking a long time to adjust to dark lighting
- Blurred vision while reading
- Metamorphopsia- a visual defect that causes lines to look curvy or round.
Additionally, Elmiron plaintiffs claim that the medication caused them to experience the following symptoms:
- Vision loss
- Blind spots in their vision
- Dark spots in your vision
- Straight lines appear curvy
- Straight lines appear curvy
- Muted colors
- Increased sensitivity to light
- Eye color changes
- Distorted vision
- Blurred vision
How do you determine if Elmiron affected your vision?
Vision problems and using Elmiron do not directly qualify you for compensation. It requires expert examination to determine if the cause of your vision problems was due to the use of Elmiron. Evidence is critical in winning any court case. Remember, Elmiron vision problems present the same symptoms as age-related macular deterioration, which could be challenging.
Valid evidence you may be required to provide includes the following:
- Medical records, doctors’ notes, and records of bills
- Prove medical research connecting the use of Elmiron to vision problems or loss of eyesight
- Receipts or documents supporting your use of Elmiron
- Your Elmiron prescription and dosage information
You will need the services of a product liability attorney to help you prove that Elmiron manufacturer is responsible for the vision damages. In such product liability cases, liability rules apply. As a result, you need to prove that the product was defective and you used the product as intended when the harm occurred. The manufacturer is held liable regardless of whether it was a negligence case or if the harm was intended.
A drug may be defective in the following ways:
- If it has serious side effects
- Defective manufacturing
- Improper marketing
Why should you file an Elmiron Product Liability Lawsuit?
If it is established that Elmiron permanently damaged your eyesight, you would be liable for compensation by the manufacturer. The types of compensation you could get from your Elmiron personal injury claim include the following:
- Medical bills
- Loss of wages
- Emotional distress
- Pain and suffering
- Anticipated future losses related to the injury
- Punitive damages if Janssen Pharmaceuticals is found to have acted maliciously or grossly negligent
The compensation amounts you get from a product liability case depend on the severity of the damages. Those with serious injuries get higher compensation.
Pharmaceutical companies named in the Elmiron Class Action Lawsuit include
- Janssen Pharmaceuticals, owned by Johnson & Johnson
- Centocor
- Alza
- TEVA
- Bayer
- Ortho-McNeil
- IVAX
What is expected of the Elmiron Lawsuit Settlements?
Since there haven’t been any trials, it is hard to determine the expected settlements. Based on the history of other prior mass tort drug cases, experts believe that average settlements in the Elmiron litigation could be around thousands of dollars. The least is expected to be $25,000, up to $1M for 100% vision loss.